Diagrams of Ideal and Real Gas Processes - Thermodynamics
Thermodynamics Directory | Heat Transfer Directory
Diagrams of Ideal and Real Gas Processes
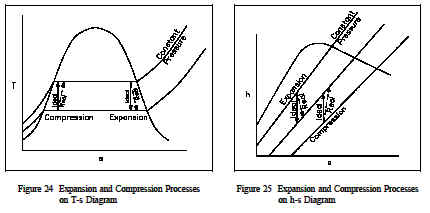
Any ideal thermodynamic process can be drawn as a path on a property diagram, such as a T-s or an h-s diagram. A real process that approximates the ideal process can also be represented on the same diagrams (usually with the use of dashed lines).
In an ideal process involving either a reversible expansion or a reversible compression, the entropy will be constant. These isentropic processes will be represented by vertical lines on either T-s or h-s diagrams, since entropy is on the horizontal axis and its value does not change. A real expansion or compression process operating between the same pressures as the ideal process will look much the same, but the dashed lines representing the real process will slant slightly towards the right since the entropy will increase from the start to the end of the process. Figures 24 and 25 show ideal and real expansion and compression processes on T-s and h-s diagrams.